TechNotes No. 12-2000
Binding of a plant lectin horseradish peroxidase
conjugate to a-D-galactoside GlycoWell™
plates.
Introduction
Binding
of lectins to immobilized saccharides and subsequent inhibition of such a binding
event is often used for evaluating lectin inhibitors. The use of GlycoWell™
plates facilitates an ELISA assay involving the binding of lectins to the wells.
This TechNote describes the binding of an a-D-galactopyranoside
recognizing lectin Maclura pomifera1, to a-D-galactopyranoside
GlycoWell™ plates.
Experimental
- Lectin incubation. Add 100 µL lectin solution to
each well. Leave for 45 min
- Washing. Empty the wells
and wash three times with CovaBuffer. Leave the last wash in the wells for 15
min. Wash with citrate/phosphate buffer once.
- Substrate
reaction. Add 100 µL substrate solution to each well. Read optical density
at 490 nm in a microwell plate reader.
Discussion
The results demonstrate that a a-D-galactopyranoside
GlycoWell™ plate efficiently binds a a-D-galactopyranoside-recognizing
plant lectin, Maclura pomifera (Figure 1). Thus, GlycoWell plates are suitable
for detecting carbohydrate binding properties of proteins.
Materials
- GlycoWell™ plates SW-00-001 (N-acetyl, blank)
and SW-01-007 (a-D-galactopyranoside).
- PBS buffer pH 7.2, 0.15 M Na+: 24.0 g NaCl, 0.6 g KCl, 3.46
g Na2HPO4·2H2O, 0.6 g KH2PO4,
3 L H2O.
- TWEEN/PBS: 0.10 mL TWEEN 20, 200 mL PBS.
CovaBuffer:
116.9 g NaCl, 10.0 g MgSO4·7H2O, 0.50 mL TWEEN 20, 1 L PBS.
- Citrate-Phosphate buffer, pH 5.0: 7.30 g citric acid, 11.86 g Na2HPO4·2H2O,
1 L H2O.
- Lectin stock: 1.0 mg Maclura pomifera-horseradish
peroxidase conjugate (SIGMA L4401), 1.0 mL TWEEN/PBS.
- Lectin solution:
70 µL Lectin stock, 7.0 mL TWEEN/PBS, filtered through a 0.22 micron filter.
-
Substrate solution: 6.0 mg o-phenylenediamine, 10.0 mL citrate/phosphate
buffer, 5 µL H2O2, freshly prepared.
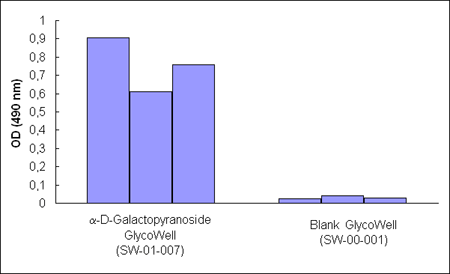 Figure
1. A Maclura pomifera-horseradish peroxidase
conjugate binds to a-D-galactopyranoside
GlycoWell™ plates (SW-01-007), but not to a GlycoWell™ plate devoid
of saccharide (SW-00-001). Figure
1. A Maclura pomifera-horseradish peroxidase
conjugate binds to a-D-galactopyranoside
GlycoWell™ plates (SW-01-007), but not to a GlycoWell™ plate devoid
of saccharide (SW-00-001). |
Reference.
1. Jones, J.M. and Feldman, J.D. J. Immunol.,
1973, 3, 1765.
 Figure
1. A Maclura pomifera-horseradish peroxidase
conjugate binds to a-D-galactopyranoside
GlycoWell™ plates (SW-01-007), but not to a GlycoWell™ plate devoid
of saccharide (SW-00-001).
Figure
1. A Maclura pomifera-horseradish peroxidase
conjugate binds to a-D-galactopyranoside
GlycoWell™ plates (SW-01-007), but not to a GlycoWell™ plate devoid
of saccharide (SW-00-001).